Development of Critical Quality Attributes as part of Antibody Drug Conjugate Control Strategy
The development of analytical control strategies for monitoring critical quality attributes (CQAs) remains one of the fundamental challenges in biotherapeutic advancement, particularly for complex modalities. Antibody-drug conjugates (ADCs) represent a sophisticated class of therapeutics where the convergence of protein science and chemical modification creates distinctive analytical hurdles that demand innovative methodological approaches.
The conjugation of cytotoxic payloads to antibodies introduces multiple parameters requiring precise characterization and monitoring—glycoprofile alterations, drug-antibody ratio (DAR) variability, conjugation site heterogeneity, unconjugated linker presence, cytotoxicity potency, and host cell protein contamination. This article examines these challenges alongside effective analytical strategies that address the complexities inherent to ADC characterization. The methodologies presented herein offer insights into overcoming persistent obstacles in establishing robust control strategies essential for consistent product quality, efficacy, and safety throughout development and manufacturing.
ADC Glycosylation and Drug Antibody Ratio (DAR)
The effectiveness or strength of an ADC is in large part determined by the load of drug attached to each antibody molecule and its retention of target binding properties. The extent of drug conjugation is controlled through process chemistry and monitored as a drug-antibody ratio (DAR). In early development, DAR is typically monitored using LC-HRMS of the intact ADC. By identifying the sites and extent of conjugation one can be assured that the ADC maintains strength and targeting.
LC-HRMS analysis can also be used to:
- Reveal conjugation of linkers without drug
- Monitor glycoforms in the ADC to ensure consistency with the precursor antibody
LC-HRMS was utilized in ADC-X analysis on a Vanquish Flex-Orbitrap 240 (Thermo Fisher Scientific) system using a BioResolve RP mAb column (Waters) for intact and subunit analysis. Data processing was performed in BioPharma Finder. ADC was injected as either intact or digested sample. For subunit analysis, the ADC was digested with IdeS (Promega) at 37°C for one hour before injection. Some of this material was subsequently incubated with 10 mM DTT in 4 M guanidine-HCl, 67 mM Tris, 1.3 mM EDTA, pH 8 at 56°C for 45 minutes.
ADC intact mass analysis deconvoluted data revealed a series of intact ADC masses consisting of glycoforms with various degrees of drug conjugation (see Figure 1). The average DAR was calculated as 3.07.
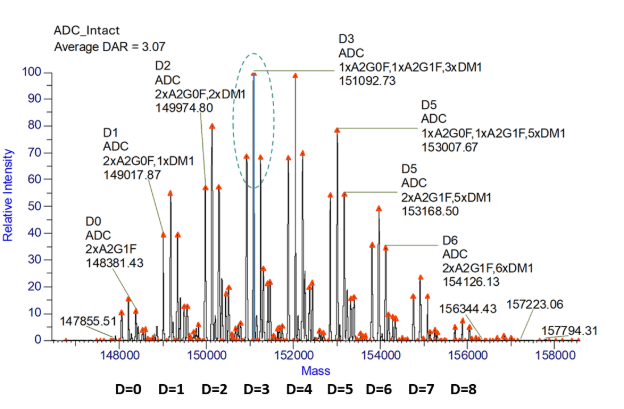
Figure 1. Intact mass analysis
The subunit analysis showed F(ab)’2 subunits with degree of drug conjugate ranging from 0 to 6. The presence of conjugated linker without drug is also evident (see Figure 3). No glycans were observed in the F(ab)’2 subunit.
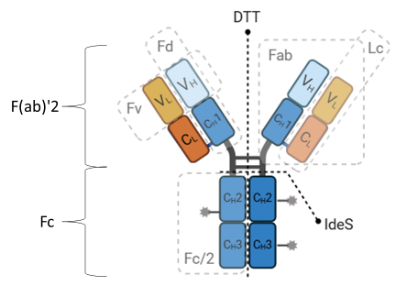
Figure 2. Illustration of an ADC and subunits upon digestion
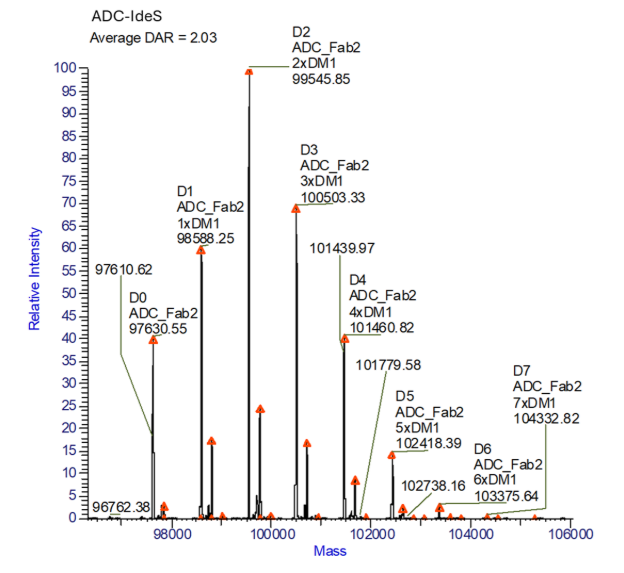
Figure 3. Subunit analysis, IdeS digestion
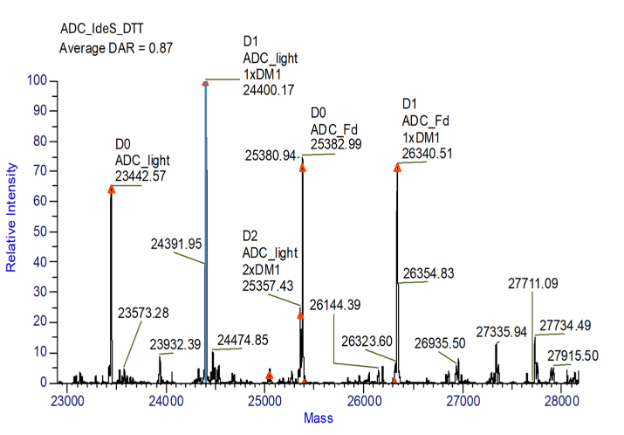
Figure 4. Subunit analysis, IdeS DTT digestion
Peptide mapping
For peptide mapping on a Waters Acquity UPLC Peptide BEH C18 column, intact ADC was digested with SMART Digest soluble trypsin prior to analysis of tryptic peptides. Data processing was performed in BioPharma finder.
Peptide mapping yielded complete coverage of the ADC (see Figure 5) and revealed amino acids carrying drug conjugated, as evidenced by a predicted mass shift and a signature drug fragmentation ion (see Figure 6). Attachment of drug was confirmed by liquid chromatography tandem mass spectrometry (LC-MS/MS) for 4 lysine residues, namely K42, K149, K88, K217).
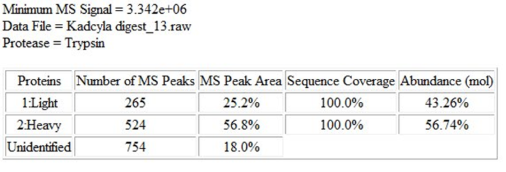
Figure 5. Sequence coverage map
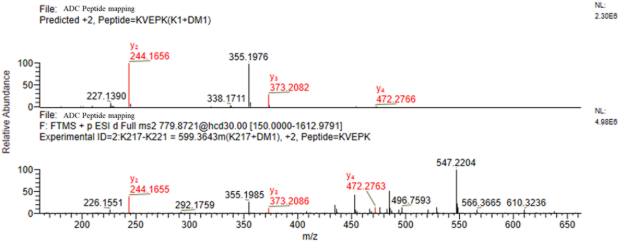
Figure 6. Peptide mapping
Potency
Potency of a drug is determined by using a measurable result that can be directly attributed to the function of that drug. For ADCs, the mechanism of action typically involves cell killing, which is often measured by either antibody-dependent cellular cytotoxicity (ADCC) or cell dependent cytotoxicity (CDC) monitoring.
While binding assays may be sufficient for meeting regulatory requirements, cell-based potency assays are the preferred approach for later phases. Key considerations in the development of a cell-based potency assay include:
- Identifying biologically relevant cells
- Engineering cell lines or co-culture systems
- Strategizing dosing and curve fitting to develop repeatable and reliable assays
The final activity read out platform for cell-based potency assays may vary. It may be worthwhile considering kit-based approaches such as measuring targeted cancer cell death using Cell Titer-Blue assay system or using engineering cells with NFAT luciferase reports driving expression of luciferase (see Figure 7).
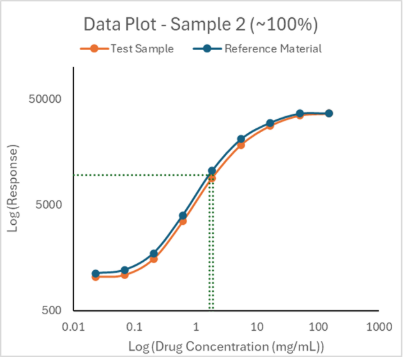
Figure 7. ADCC relative potency assay
Quantity of host cell protein
Residual DNA and host cell protein (HCP) control is critical at the drug intermediate level due to impurities introduced during the cell culture stages. The amount of process-related impurities can be empirically determined using various platform approaches. During the early phases of development, commercially available HCP kits may be a suitable option. For later phase products, however, product-specific methods will be required.
Meso Scale Discovery (MSD) assays developed using antibodies against product specific HCPs can detect and quantify levels of existing impurities using simple enzyme-linked immunosorbent assay (ELISA) techniques. The precise amount of HCPs can be determined by comparing to a reference standard (see Figure 8).
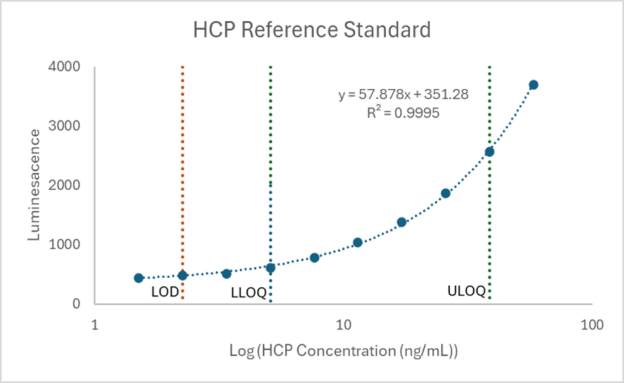
Figure 8. HCP reference standard curve
Key takeaway
There are certain CQAs that should be monitored during the development and manufacturing of any ADC. By focusing on core CQAs—including glycan analysis, potency, and impurities assays—and determining correlations among them, ADC developers can better control the complex nature of these biologics, address potential safety and efficacy concerns, and streamline the development process from early phases to regulatory approval and commercial production.
To learn more about developing and executing a robust ADC control strategy, speak to one of our scientists.

Development of Critical Quality Attributes as part of Antibody Drug Conjugate Control Strategy