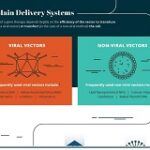
Gene Therapy
Immune responses to AAV vectors: overcoming barriers to successful gene therapy
Mingozzi F, High KA. Immune responses to AAV vectors: overcoming barriers to successful gene therapy. Blood. 2013;122:23-36. Abstract Gene therapy products for the treatment of…