Immunogenicity
Progress and promise of pharmacodynamic biomarkers: novel strategies and assay considerations in drug development
Pharmacodynamic (PD) biomarkers provide crucial insights into a drug’s mechanism of action (MoA) and efficacy by measuring its effects on biological targets within an organism.…
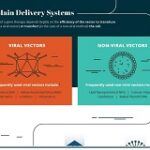
Technical Considerations of Pharmacokinetic Assays for LNP‑mRNA Drug Products by RT‑qPCR
Lipid nanoparticle-messenger RNA (LNP-mRNA) drug products are a growing class of drug modalities. The unique composition of these drug products requires multiple measurements to account…
Why is Biomarker Assay Validation Different from that of Pharmacokinetic Assays?
The Food and Drug Administration (FDA) issued a long awaited guidance document on Bioanalytical Method Validation for Biomarkers (BMVB) (1) in January 2025, which replaces…
Assessment of Neutralizing Antibody Activity in Clinical Studies: Use of Surrogate Measurements Instead of Stand‑alone Assays
Neutralizing antibodies (NAbs) to protein therapeutics have traditionally been assumed to be the most impactful subset ofanti-drug-antibodies (ADA). NAbs can block the biotherapeutic from engaging…
Development of a validated novel bead extraction method for the detection of anti-PEG antibodies in human serum
Polyethylene glycol (PEG) is used in many applications including drug development. Due to exposure to environmental products, there is a high prevalence of preexisting anti-PEG…
Managing unwanted immunogenicity of therapeutic proteins
Deehan M, Garcês S, Kramer D, Baker M, Rat D, Roettger Y, Kromminga A. Managing unwanted immunogenicity of therapeutic proteins. Autoimmunity Rev. 2015;14,569-574. Abstract All…