Protecting and Managing Your Samples to Safeguard Accurate Bioanalytical Results
As a GxP (GLP, GMP, and GCP) laboratory, BioAgilytix upholds sample management methodologies to ensure your sample analysis results are reliable, reproducible, and compliant.
A laboratory’s bioanalytical services can only be as good as the quality with which they manage and protect samples. Therefore, an integrated approach to sample management is an essential control required to uphold a commitment to outstanding bioanalytical quality. Ensuring timely, accurate analysis of your collected samples, BioAgilytix implements rigorous sample management procedures to mitigate variability and quality concerns. Trust your samples in our expert hands.Learn More About Our Sample Management Processes:
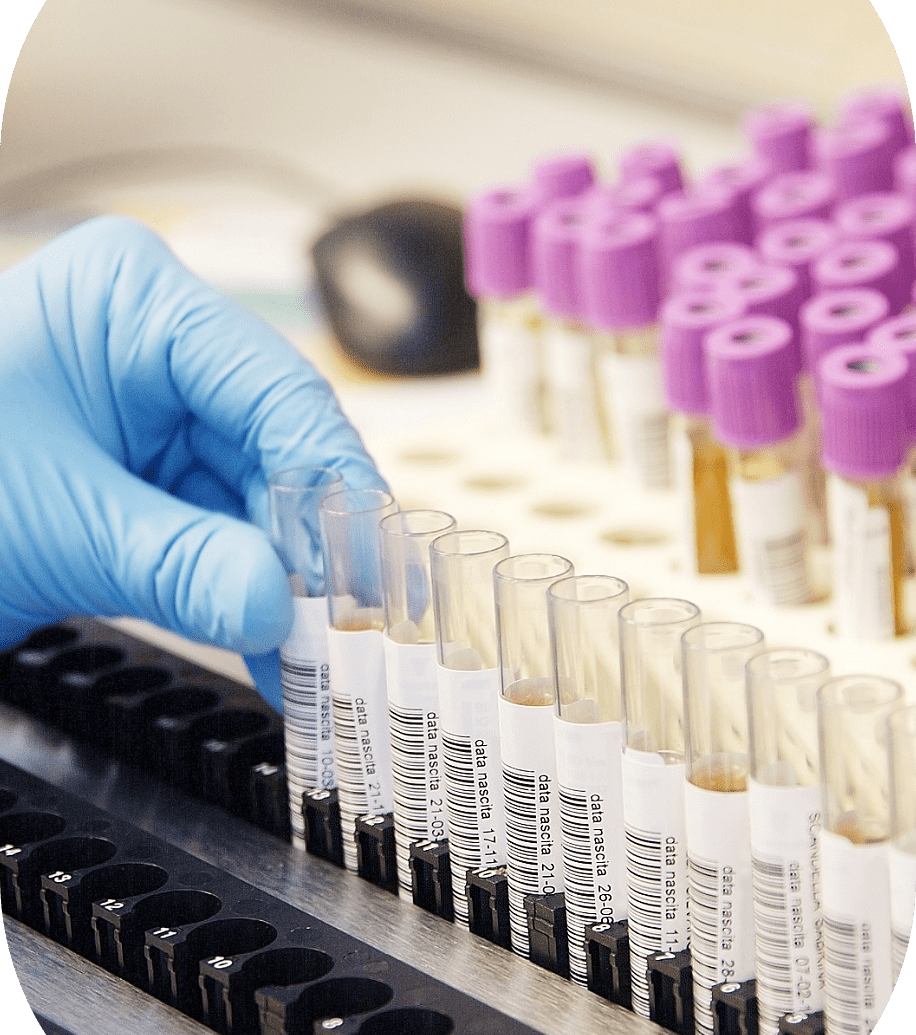
Upholding the Four Key Pillars of Sample Management
BioAgilytix understands that our customers are trusting us with precious samples, and that we must uphold their integrity to ensure accurate analysis and reliable results. As such, we have processes in place to ensure we effectively address the four key areas of effective sample management:
- Sample Handling Accuracy: In order to ensure the accurate handling of all samples at every stage of development, sample management procedures must be in place for clear and consistent sample processing and labeling. This pillar of sample management is paramount in order to drive scientific excellence.
- Sample Analysis: Mitigating variability and concerns of compromised sample integrity requires that your collected samples be analyzed efficiently and in a timely manner. The timeliness of sample analysis is paramount to achieving maximum reliability and accuracy.
- Sample Protection: Protecting samples involves more than keeping samples secure. It also means maintaining sample integrity through the properly regulated and monitored storage, and clearly defined processes for their movement and retrieval throughout in the laboratory environment.
- Sample Procedures: Of course, each pillar of sample management must be carried forth consistently and effectively in the laboratory environment. Therefore, sample management has an integral procedural pillar. By implementing standard sample control and operating procedures at every stage of the development process, BioAgilytix ensures that results from analyzed samples are reliable and reproducible.

Rely On Consistent Sample Management Process
As a trusted GxP laboratory partner to our clients, we understand that your samples are of critical importance, and have multiple processes in place to guarantee their optimal management and protection, including:
- Samples accessioned using a fully validated, electronic system
- Control process in place for sample movement and sample retrieval throughout the lab
- Refrigerators and freezers on automated monitoring system 24/7